Synergistic impacts of sonolysis aided photocatalytic degradation of water pollutant over perovskite-type CeNiO3 nanospheres†
Abstract
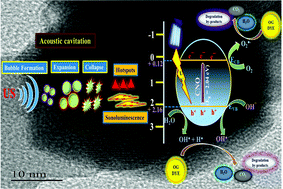
The current study reports on the preparation of perovskite-type CeNiO3 nanostructures as a sonophotocatalyst via a facile hydrothermal approach followed by annealing at 800 °C. The CeNiO3 catalyst was characterized by various physicochemical techniques to comprehend the crystallinity, microstructure, and morphological features of perovskite nanostructure. Subsequently, the sonophotocatalytic (a hybrid technique involving sonolysis and photocatalysis) performance was assessed for the removal of Orange-G (OG) dye over the surface of CeNiO3 (CNO) perovskite from aqueous media under 4 h. The degradation efficacy of OG followed the sequence: photocatalysis < sonocatalysis < sonophotocatalysis. The optimal experimental conditions (ultrasound frequency: 160 kHz, UV light intensity: 6 W, catalyst dosage: 0.2 g L−1, and OG concentration: 2 × 10−5 mol L−1) displayed remarkable sonophotocatalytic efficacy on the decomposition of OG, with a removal proficiency of ∼95% and the reaction followed pseudo-first-order rate kinetics. A possible mechanism for the sonophotocatalytic degradation of OG has been proposed.


 Please wait while we load your content...
Please wait while we load your content...