Charge transfer in mixed and segregated stacks of tetrathiafulvalene, tetrathianaphthalene and naphthalene diimide: a structural, spectroscopic and computational study†‡
Abstract
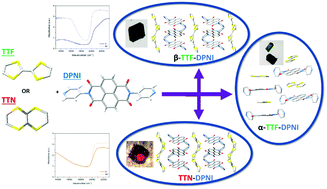
Tetrathiafulvalene (TTF) is a highly tunable electron donor that has been widely studied in charge transfer (CT) complexes, including Bechgaard salts which are superconductors at low temperatures. Its close relative, tetrathianaphthalene (TTN) has received considerably less attention than its TTF counterpart but is potentially a versatile electron donor. Three novel CT complexes containing mixed and segregated stack donor-acceptor (D–A) complexes of tetrathiafulvalene (TTF), tetrathianaphthalene (TTN) and naphthalene diimide (NDI) are reported. We demonstrate a facile mechanochemical synthesis of two salts, TTF–DPNI and TTN–DPNI (DPNI = N,N′-di-(4-pyridyl)-1,4,5,8-naphthalenetetracarboxydiimide), which is uncommon for these materials. Despite the detection of a very low partial charge transfer in the three salts using UV-Vis-NIR, Raman and EPR spectroscopies, we elucidated the properties of the TTN core with support from DFT calculations for the first time. We highlight the performance of TTN in a CT complex and demonstrate the importance of a combined approach to the characterisation of CT in organic D–A complexes.


 Please wait while we load your content...
Please wait while we load your content...