Structural diversity and magnetic properties of copper(ii) quinaldinate compounds with amino alcohols†
Abstract
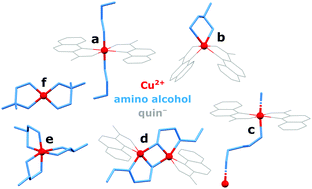
The reactions between [Cu(quin)2(H2O)] (quin− = the anionic form of quinoline-2-carboxylic acid) and a series of aliphatic amino alcohols have yielded structurally very diverse copper(II) complexes, labelled a–g. Single-crystal X-ray structure analysis has revealed either intact amino alcohol molecules or amino alcoholate ions serving as ligands. In type a complexes, the amino alcohols are bound in a monodentate manner via NH2. Engagement of both functional groups in coordination was observed for types b and e (a bidentate chelating mode) and type c (a bidentate bridging one) complexes. In view of the strong bidentate chelating coordination of quinaldinate in [Cu(quin)2(H2O)], the formation of homoleptic amino alcohol complexes e was not anticipated. Equally surprising was the transformation of a mononuclear starting material into a one-dimensional (1D) coordination polymer, [Cu(quin)2]n (g). Spontaneous deprotonation of some amino alcohols and coordination of, thus formed, amino alcoholates via both donors also took place. Dinuclear complexes (d) contained two bridging amino alcoholates, whilst bidentate chelating mode was observed for type f. Interestingly, the dinuclear complex exists as two isomers which differ in the position of quinaldinates with respect to the Cu(μ-OR)2Cu core. DFT calculations on isolated syn- and anti-[Cu2(quin)2(3a1pO)2] (3a1pO− = anion of 3-amino-1-propanol) have shown the syn isomer to be more stable. The explanation lies in the intramolecular π⋯π stacking of quinaldinates, possible only in this isomer. Magnetic susceptibility measurements revealed antiferromagnetic interactions between S = 1/2 copper(II) spins in all the studied compounds except in [Cu(quin)2]n (g) for which weak ferromagnetic couplings are detected.


 Please wait while we load your content...
Please wait while we load your content...