Two-phase synthesis of Fe doped cerium phosphate ultra-fine nanocrystals for efficient oxygen evolution†
Abstract
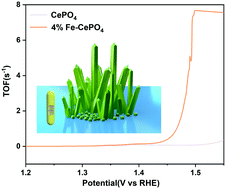
It is very important and challenging to develop electrocatalysts with high performance and economic benefits. In this work, ultra-fine Fe doped CePO4 nanocrystals (Fe–CePO4) were prepared by a simple two-step hydrothermal synthesis method under mild conditions, combining the advantages of the PO43− group, easy hydroxylation of metal sites, convenient oxygen ion transfer due to the polyvalent nature of cerium ions, and a high charge transfer rate due to the doping of the second metal. The combination of these advantages improves the conductivity of the Fe–CePO4 electrocatalyst, makes it exhibit a faster charge transfer ability and shows excellent OER performance, with only 197 mV and 253 mV overpotentials at current densities of 10 mA cm−2 and 100 mA cm−2, an ultra-small Tafel slope of 52.4 mV dec−1 and a high turnover frequency (TOF) of 7.67 s−1 at 1.50 V. This synthesis method of ultrafine nanocrystals and the combination of the advantages of various groups in this work provide ideas and insights for the subsequent preparation of efficient and economical electrocatalysts.


 Please wait while we load your content...
Please wait while we load your content...