Sustainable synthesis of bio-based PTMEG and methane beyond the Reppe method†
Abstract
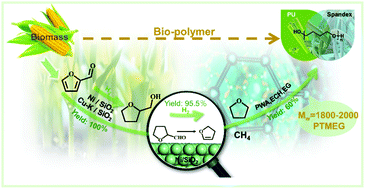
To resolve the issues of a highly polluting and energy-intensive industry based on the Reppe method, herein we developed an economically feasible green route for the synthesis of poly(tetramethylene ether glycol) (PTMEG) and methane from furfural (FA). The synthesis route consisted of the base metal Ni and/or Cu catalyzed full hydrogenation of FA, followed by the subsequent selective hydrodeoxygenation of tetrahydrofurfuryl alcohol (THFA) to tetrahydrofuran (THF) and methane. The latter was the key step for the process, where tetrahydrofurfural acted as an important intermediate in the alcohol–aldehyde equilibrium of THFA conversion on the Ni surface in a fix-bed reactor. This was followed by its decarbonylation to 2,3-dihydrofuran and CO, and subsequent hydrogenation to THF and methane. THF polymerization was catalyzed by phosphotungstic acid (PWA) with epichlorohydrin (ECH) as the initiator to produce PTMEG with a controllable molecular weight and distribution (Mw = 1889, D = 1.483). The comparative techno-economic analysis also demonstrated that the furfural method was more responsive to economic and green concerns than the traditional Reppe technique. Therefore, as the highly polluting and energy-intensive Reppe industry will most likely be restricted in a low carbon society, the green and economical furfural method is poised to establish sustained competitiveness for producing bio-PTMEG and bio-methane in the future.


 Please wait while we load your content...
Please wait while we load your content...