Ultrasonic-assisted oxidation of cellulose to oxalic acid over gold nanoparticles supported on iron-oxide†
Abstract
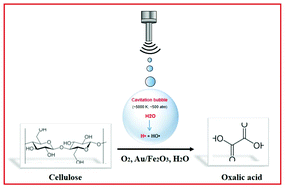
The use of unconventional activation techniques, such as low frequency ultrasound (US), in combination with heterogeneous catalysts offers a powerful synergistic approach to transform renewable resources to value added chemicals. In this context, we report a catalytic base-free strategy for the selective oxidation of microcrystalline cellulose to oxalic acid (OA) by combining low frequency ultrasound and Au/Fe2O3 as a catalyst. We demonstrate that low frequency ultrasound induces the fragmentation of cellulose particles, making it more prone to catalytic oxidation in the presence of Au/Fe2O3. Under the optimized conditions, OA was obtained in 45% yield in the presence of molecular oxygen, corresponding to an overall yield of 53% into carboxylic acids (gluconic, formic, 2-keto-gluconic acid, etc.). Furthermore, by means of density functional theory, it was demonstrated that a charge transfer occurred from Au nanoparticles to Fe2O3, resulting in the formation of active catalytic species capable of decomposing H2O2, formed by sonolysis of water, to reactive O* species that were involved in the oxidation of cellulose. This charge transfer was also highlighted by X-ray photoelectron spectroscopy which revealed a partial oxidation of Au0 to Au3+.
- This article is part of the themed collection: 2022 Green Chemistry Hot Articles


 Please wait while we load your content...
Please wait while we load your content...