Mild and efficient photocatalytic coupled alcohol oxidative dehydrogenation and CO2 reduction driven by visible light†
Abstract
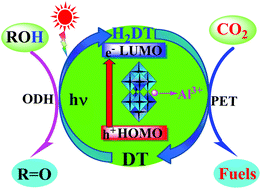
The advanced coupled conversion technique based on the oxidative dehydrogenation (ODH) of alcohols and the reduction of CO2 is a potential strategy to solve the current energy and environmental crisis, and is of great significance for the upgrading conversion of CO2 to fuels. This work discloses that the Al3+ species-doped decatungstates (Al-DTs) can efficiently catalyze the coupled conversion of various aliphatic and aromatic primary/secondary alcohols with CO2 in MeCN under visible light illumination and very mild conditions, and the reductive efficiency of CO2 depends on the doping levels of Al3+ species in Al-DTs and the ODH reactivity of alcohols. After continuously illuminating for 12 h, the best, 4% Al-DT can provide 437 μmol acetophenone as the ODH product and 218 μmol CO2 reduction products (formic acid and methanol) with 1.82 h−1 turnover frequency in the photocatalytic coupled conversion of α-phenylethanol with CO2. Also, a trace amount of CO is detected from the photocatalytic coupled reactions. UV-vis spectra and isotope tracing tests using heavy oxygen water (H218O) support that the doubly-reduced blue H2DT species generated from the photocatalytic ODH of alcohols turns into its oxygen-deficient form ([DT2−]v) via an intra-molecular dehydration or lattice oxygen transfer and the latter, with the help of doped Al3+ species, may efficiently reduce CO2 to fuels via the multi-step proton-coupled electron transfer (PET) pathway.


 Please wait while we load your content...
Please wait while we load your content...