Superlattice ordering transitions driven by short-range structure in barium calcium carbonates
Abstract
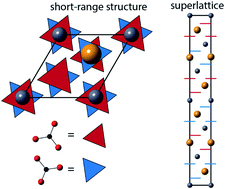
Balcite (BaxCa1−xCO3) is a synthetic analog of rhombohedral carbonate minerals like calcite and dolomite that is disordered on both the cation and anion sublattices. Here, we show that multiple exotic superlattice structures, including a dolomite analog that we call balcomite, can form from balcite at elevated temperatures. The second-order balcite-to-balcomite conversion at temperatures between 150–600 °C is driven by the preference of barium and calcium for different oxygen coordination numbers and facilitated by local carbonate reorientation. At elevated pressure, further superlattice order arises from cation segregation in all three dimensions, producing a supercell with the same R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m symmetry as balcite but 6× larger. This highly ordered structure relaxes back to the balcomite structure upon returning to ambient conditions. None of the three naturally occurring polymorphs of Ba0.5Ca0.5CO3 (alstonite, paralstonite, barytocalcite) formed from balcite despite being putatively energetically favored. Instead, alstonite transformed to a balcomite-like structure via a first-order process after transiently converting to a paralstonite-like structure via a second-order process. Together, these results show that high temperature transformation pathways between structures in the barium calcium carbonate system can be driven by coarsening and are facilitated by similarity in short-range order, conceptually analogous to previously described low-temperature transformations. Many of the exotic high temperature carbonate structures are unstable, but may participate in transformation pathways between naturally observed metastable mineral phases, suggesting important roles for ephemeral phases in shaping past and current mineral distributions.
m symmetry as balcite but 6× larger. This highly ordered structure relaxes back to the balcomite structure upon returning to ambient conditions. None of the three naturally occurring polymorphs of Ba0.5Ca0.5CO3 (alstonite, paralstonite, barytocalcite) formed from balcite despite being putatively energetically favored. Instead, alstonite transformed to a balcomite-like structure via a first-order process after transiently converting to a paralstonite-like structure via a second-order process. Together, these results show that high temperature transformation pathways between structures in the barium calcium carbonate system can be driven by coarsening and are facilitated by similarity in short-range order, conceptually analogous to previously described low-temperature transformations. Many of the exotic high temperature carbonate structures are unstable, but may participate in transformation pathways between naturally observed metastable mineral phases, suggesting important roles for ephemeral phases in shaping past and current mineral distributions.
- This article is part of the themed collection: Understanding Crystallisation


 Please wait while we load your content...
Please wait while we load your content...