Formation sequence of solid electrolyte interphases and impacts on lithium deposition and dissolution on copper: an in situ atomic force microscopic study†
Abstract
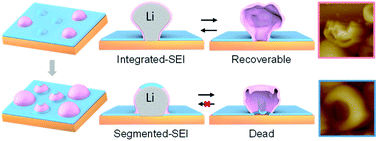
Copper is the most widely used substrate for Li deposition and dissolution in lithium metal anodes, which is complicated by the formation of solid electrolyte interphases (SEIs), whose physical and chemical properties can affect Li deposition and dissolution significantly. However, initial Li nucleation and growth on bare Cu creates Li nuclei that only partially cover the Cu surface so that SEI formation could proceed not only on Li nuclei but also on the bare region of the Cu surface with different kinetics, which may affect the follow-up processes distinctively. In this paper, we employ in situ atomic force microscopy (AFM), together with X-ray photoelectron spectroscopy (XPS), to investigate how SEIs formed on a Cu surface, without Li participation, and on the surface of growing Li nuclei, with Li participation, affect the components and structures of the SEIs, and how the formation sequence of the two kinds of SEIs, along with Li deposition, affect subsequent dissolution and re-deposition processes in a pyrrolidinium-based ionic liquid electrolyte containing a small amount of water. Nanoscale in situ AFM observations show that sphere-like Li deposits may have differently conditioned SEI-shells, depending on whether Li nucleation is preceded by the formation of the SEI on Cu. Models of integrated-SEI shells and segmented-SEI shells are proposed to describe SEI shells formed on Li nuclei and SEI shells sequentially formed on Cu and then on Li nuclei, respectively. “Top-dissolution” is observed for both types of shelled Li deposits, but the integrated-SEI shells only show wrinkles, which can be recovered upon Li re-deposition, while the segmented-SEI shells are apparently top-opened due to mechanical stresses introduced at the junctions of the top regions and become “dead” SEIs, which forces subsequent Li nucleation and growth in the interstice of the dead SEIs. Our work provides insights into the impact mechanism of SEIs on the initial stage Li deposition and dissolution on foreign substrates, revealing that SEIs could be more influential on Li dissolution and that the spatial integration of SEI shells on Li deposits is important to improving the reversibility of deposition and dissolution cycling.
- This article is part of the themed collection: Next Generation Nanoelectrochemistry


 Please wait while we load your content...
Please wait while we load your content...