A trade-off between ligand and strain effects optimizes the oxygen reduction activity of Pt alloys†
Abstract
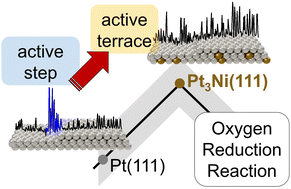
To optimize the performance of catalytic materials, it is paramount to elucidate the dependence of the chemical reactivity on the atomic arrangement of the catalyst surface. Therefore, identifying the nature of the active sites that provide optimal binding of reaction intermediates is the first step toward a rational catalyst design. In this work, we focus on the oxygen reduction reaction (ORR), an essential constituent of several energy provision and storage devices. Among the state-of-the-art ORR catalysts are platinum (Pt) and its alloys. The latter benefit from the so-called ligand and strain effects, which influence the electronic properties of the surface. Here, we “visualize” the active sites on Pt3Ni(111) in an acidic medium with a lateral resolution in the nanometre regime via an in situ technique based on electrochemical scanning tunnelling microscopy. In contrast to pure Pt, where the active sites are located at concave sites close to steps, Pt3Ni(111) terraces contain the most active centres, while steps show activity to a comparable or lesser extent. We confirm the experimental findings by a model based on alloy- and strain-sensitive generalized coordination numbers. With this model, we are also able to assess both the composition and the geometric configuration of optimal catalytic active sites on various Pt alloy catalysts. In general, the interplay of ligand effects and lattice compression resulting from the alloying of Pt with 3d transition metals (Ti, Co, Ni, Cu) gradually increases the generalized coordination number of surface Pt atoms, thereby making (111) terraces highly active. This combination of theoretical and experimental tools provides clear strategies to design more efficient Pt alloy electrocatalysts for oxygen reduction.


 Please wait while we load your content...
Please wait while we load your content...