How membrane characteristics influence the performance of CO2 and CO electrolysis†
Abstract
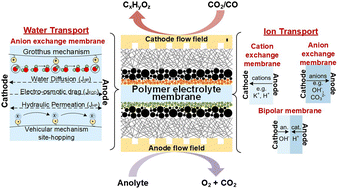
Due to the ability to produce sustainably carbon-based chemicals and fuels, CO2 electrolysis and the closely related CO electrolysis are advancing rapidly from fundamental studies toward industrial applications. Many near-room temperature CO2 and CO electrolysis (CO(2)E) technologies adopt features from proton exchange membrane fuel cells and H2 electrolyzers. However, CO(2)E's selectivity and overall performance are highly sensitive to a multitude of parameters, adding an extra degree of complexity. One often-overlooked parameter in optimizing these devices is the ion exchange membranes (IEM). Here we critically review the IEM performance variables of most relevance to CO(2)E, which leads to identifying several parameters in need of substantial more scientific understanding. We begin with a summary of the working principles of the three main IEM types for CO(2)E, then focus on anion exchange membranes (AEM) since AEMs provide the most favorable local alkaline environment for CO(2)E at the cathode. Critical issues for AEMs in CO2E include (i) ion and water transport in the membrane, (ii) ionic conductivity, and (iii) chemical stability. We conclude with an overview of the state-of-the-art IEM reported in high current density (j ≥ 100 mA cm−2) CO2 and CO electrolysis devices.


 Please wait while we load your content...
Please wait while we load your content...