A Zn–nitrite battery as an energy-output electrocatalytic system for high-efficiency ammonia synthesis using carbon-doped cobalt oxide nanotubes†
Abstract
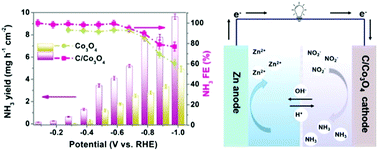
Ammonia (NH3) is a carbon-free fuel and essential for producing chemicals like fertilizers. The conversion of toxic nitrite (NO2−) ions from contaminated water to NH3 in an electrocatalytic system is highly attractive both environmentally and sustainably and the system can potentially be changed from energy input to energy output. In this work, we develop a Zn–NO2− battery meanwhile working as an electrocatalytic system that can simultaneously degrade NO2−, produce NH3 and generate electricity. Nanoparticle-assembled carbon-doped cobalt oxide (C/Co3O4) hollow nanotubes are found to be highly efficient for the NO2− reduction reaction (NO2−RR), which shows a faradaic efficiency (FE) of nearly 100% for NH3 production in a wide potential window from −0.1 V to −0.6 V versus a reversible hydrogen electrode. The interstitial C dopant can induce a local electric field to greatly fascinate charge transfer, which lowers the energy barrier of the rate-determining step (*N + e− + H2O → *NH + OH−), facilitating the NO2−RR process. The assembled Zn–NO2− battery exhibits a power density of 6.03 mW cm−2 and a highest NH3 FE of 95.1%. This work not only initiates the Zn–NO2− battery system for sustainable energy supply and NH3 electrosynthesis but also offers an attractive electrocatalytic approach to realize the degradation of the NO2− pollutant in wastewater.


 Please wait while we load your content...
Please wait while we load your content...