Pushing steric limits in osmium(iv) tetraaryl complexes†
Abstract
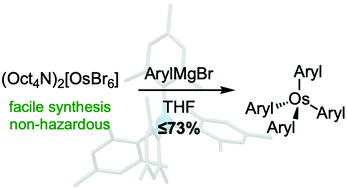
Investigations into the reactivity, properties, and applications of osmium(IV) tetraaryl complexes have been hampered by their low yielding syntheses from volatile and toxic OsO4 (typically ≤34%). Here we show that known air-stable M(aryl)4 compounds (M = Os, Ru; aryl = 2-tolyl, 2,5-xylyl) can be prepared in ≤73% yields using new, less hazardous (Oct4N)2[MX6] precursors (M = Os, Ru; X = Cl, Br). This approach also facilitates the preparation of Os(mesityl)4 (Os3) for the first time, a complex comprising bulky 2,6-dimethyl substituted aryl ligands, albeit in low yield (5%). To better understand these yield extremes, we track, by synthesizing two additional new complexes with different 2-substituted σ-aryl ligands, a clear relationship between the yields of Os(aryl)4 and ligand steric bulk. Single-crystal X-ray structures of these compounds indicate that the observed yield trend reflects the ease of accommodating aryl substituents into an open pocket that lies directly opposite each M–aryl coordination site. We perform variable-temperature 1H NMR studies of Os3, utilize a “tetrahedricity” metric to assess geometric distortion in Ru(aryl)4 and Os(aryl)4 materials, and calculate cone angle and percentage buried volume metrics to further illustrate and help quantify σ-aryl ligand steric properties. Solution cyclic voltammograms of Os(aryl)4 show that the potentials of their reversible 1−/0 and 0/1+ redox features can be fine-tuned by varying aryl substituents, and that Os3 exhibits an additional 1+/2+ redox event not previously observed in this class of compounds. Taken together, this work helps to advance the potential application of these relatively underexplored organometallic complexes in established and emerging areas of molecular materials science, such as extended molecular frameworks and self-assembled monolayers, where analogous tetraphenylmethane and silane species (M = C, Si) have been frequently targeted.


 Please wait while we load your content...
Please wait while we load your content...