π-Carbazolyl supported bis(alkyl) complexes of Sc, Y and La for α-olefin polymerization and hydrogenation†
Abstract
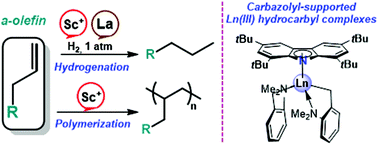
A series of new half-sandwich bis(alkyl) rare-earth metal complexes coordinated by a sterically demanding 1,3,6,8-tetra-tert-butyl-carbazol-9-yl ligand [tBu4Carb]La(CH2C6H5)2(THF) (1-La), [tBu4Carb]Ln(o-NMe2C6H4CH2)2 (Ln = Sc (2-Sc), Y (2-Y), La (2-La), [tBu4Carb]Ln(CH2SiMe3)2(THF) (Ln = Sc (3-Sc), Y (3-Y)), were synthesized. 1-La, 2-La, and 2-Y were prepared by an alkane elimination protocol, while 2-Sc, 3-Sc, and 3-Y became accessible only when salt metathesis reactions of tBu4CarbK with R2Ln(THF)n+[BPh4]− were employed. X-ray analysis revealed that in all complexes the carbazolyl ligand exhibits π-coordination with metal ions. 2-Sc and 3-Sc when activated with [Ph3C][B(C6F5)4] demonstrate excellent activity in α-olefin (octene-1, nonene-1, decene-1 and 1,1-diphenyl-but-1-ene) polymerization. When H2 was used as a chain transfer agent (1 bar, rt) in the presence of 3-Sc/[Ph3C][B(C6F5)4] or 2-Y, 2-La olefin hydrogenation occurred with quantitative conversion.


 Please wait while we load your content...
Please wait while we load your content...