The impact of secondary coordination sphere engineering on water oxidation reactivity catalysed by molecular ruthenium complexes: a next-generation approach to develop advanced catalysts
Abstract
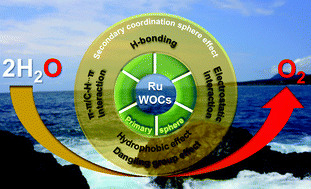
Water oxidation is the bottleneck for producing hydrogen from the water-splitting reaction. Developing efficient water oxidation catalysts (WOCs) has recently been of paramount interest. Ruthenium-based WOCs have gained much attention due to their enriched redox property, robust nature, and superior catalytic performances compared to other transition metal-based molecular catalysts. The performance of a catalyst is highly dependent on the design of the ligand framework. In nature, the secondary coordination sphere around the active site of a metalloenzyme plays a vital role in catalysis. This principle has been employed in the recent development of efficient catalysts. With the aid of secondary interactions, some landmark Ru-based WOCs, producing remarkable turnover frequencies (TOFs) in the order of 104 s−1, have been developed. In this account, we have discussed the underlying chemistry related to the effect of secondary interactions (such as hydrogen-bonding, π–π stacking, electrostatic interaction, hydrophobic–hydrophilic environment, etc.) on the kinetics of the water oxidation reaction catalysed by molecular Ru-complexes. The use of secondary interactions (such as π–π and C–H⋯π) in anchoring the molecular catalyst onto the solid conducting surface has also been discussed. We aim to provide a brief overview of the positive impact of outer-sphere engineering on water oxidation reactivity, which may offer guidelines for developing the next generation of advanced catalysts.
- This article is part of the themed collection: 2022 Frontier and Perspective articles


 Please wait while we load your content...
Please wait while we load your content...