Multivariate analysis of peptide-driven nucleation and growth of Au nanoparticles†
Abstract
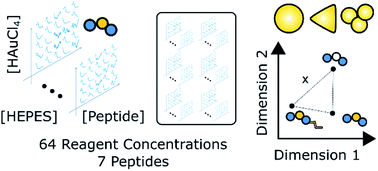
In this work we synthesize gold nanostructures using a liquid-handling robot and present new data analysis methods to quantitatively compare the effect of peptides and peptide modifications on gold nanoparticle synthesis outcomes. The peptides used were gold binding peptides Z2 and AG3, as well as five Z2 variants obtained by sequence modification or conjugation of a lipid tail. Four different concentrations of peptide (0, 0.04, 0.0894, and 0.2 mM), reducing agent (4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid, HEPES; 0, 0.01, 0.1, and 1 mM), and precursor (HAuCl4; 0.04, 0.0684, 0.117, and 0.2 mM) were used to synthesize 64 different reagent combinations for each of the seven peptides. Each sample was characterized using UV-Vis spectroscopy, which serves as a proxy for changes in nanoparticle structure, and enabled comparisons of how peptide modifications and reagent conditions affect synthesis outcomes. We then used functional data analysis to extract a pairwise signal correlation distance between each of the peptides. The signal correlation distance quantified how different the set of 64 spectra, and therefore the synthesis outcomes, was from one peptide to the other. Spherical nanoparticles synthesized in the presence of peptides were typically 10 to 50 nm in diameter, but the size and shape of nanoparticles varied with peptide properties and reagent conditions. We show that substitution of methionine for isoleucine in Z2 has a profound impact on synthesis outcomes when compared with conjugation of a lipid tail. Electron microscopy and ultra-small angle X-ray scattering were used to corroborate our conclusions from spectroscopy experiments by directly characterizing the structure of a smaller set of samples. Scanning electron microscopy revealed interconnected plate-like structures in samples prepared with a Z2 variant with all methionines substituted by isoleucine. Z2 peptides modified with a lipid tail on the other hand, formed nanoparticles which were more colloidally stable than those prepared with non-lipidated peptides, contained no plate-like particles, and whose size depended on the ratio of peptide to precursor.


 Please wait while we load your content...
Please wait while we load your content...