The versatility of non-heme diiron monooxygenase PmlABCDEF: a single biocatalyst for a plethora of oxygenation reactions†
Abstract
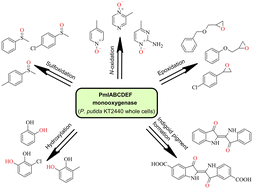
In this paper, we discuss the application of the non-heme diiron monooxygenase PmlABCDEF and its mutants in a variety of different oxidation reactions. Whole cells of Pseudomonas putida KT2440 bearing PmlABCDEF monooxygenase and its variants were shown to be versatile biocatalysts for a ring-hydroxylation, heteroatom oxidation, and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds functionalization. N-Heteroaromatic compounds such as pyridines, pyrazines, and pyrimidines were transformed to corresponding N-oxides in high substrate loads and, in several cases, with good regioselectivity. Aromatic thioethers were converted into sulfoxides as the main products of the reaction. Biooxygenation of indoles resulted in the formation of indigoid pigments, namely indigo derivatives. Conversion of aromatic compounds with substituents containing a double bond (such as styrenes and allylic ethers) produced epoxide derivatives (oxiranes). Highly valued compounds including benzyl glycidyl ether and phenyl glycidyl ether were prepared with pronounced enantiomeric excess – 93% and 77% respectively. Phenolic compounds underwent ortho-directed aromatic ring hydroxylation reactions, yielding catechol derivatives such as 3-methylcatechol, 3-ethylcatechol, and 3-chlorocatechol. Overall, we demonstrated that (1) PmlABCDEF monooxygenase is capable of transforming a vast variety of compounds into different oxy-derivatives, (2) in certain cases with high regio- or enantioselectivity, and (3) that the reached productivity titre qualifies using this biocatalyst as an alternative to chemical oxidizers for laboratory-scale preparations.
C bonds functionalization. N-Heteroaromatic compounds such as pyridines, pyrazines, and pyrimidines were transformed to corresponding N-oxides in high substrate loads and, in several cases, with good regioselectivity. Aromatic thioethers were converted into sulfoxides as the main products of the reaction. Biooxygenation of indoles resulted in the formation of indigoid pigments, namely indigo derivatives. Conversion of aromatic compounds with substituents containing a double bond (such as styrenes and allylic ethers) produced epoxide derivatives (oxiranes). Highly valued compounds including benzyl glycidyl ether and phenyl glycidyl ether were prepared with pronounced enantiomeric excess – 93% and 77% respectively. Phenolic compounds underwent ortho-directed aromatic ring hydroxylation reactions, yielding catechol derivatives such as 3-methylcatechol, 3-ethylcatechol, and 3-chlorocatechol. Overall, we demonstrated that (1) PmlABCDEF monooxygenase is capable of transforming a vast variety of compounds into different oxy-derivatives, (2) in certain cases with high regio- or enantioselectivity, and (3) that the reached productivity titre qualifies using this biocatalyst as an alternative to chemical oxidizers for laboratory-scale preparations.
- This article is part of the themed collection: Biocatalysis: A cross-journal collection


 Please wait while we load your content...
Please wait while we load your content...