Nickel carbide (Ni3C) nanoparticles for catalytic hydrogenation of model compounds in solvent†
Abstract
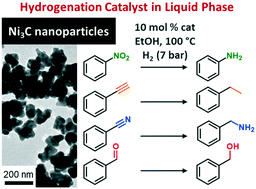
Finely divided nickel is one of the main heterogeneous catalysts for liquid phase hydrogenation of nitro and alkyne moieties. However, it constitutes a major safety hazard due to its pyrophoric character and may be subject to poisoning issues, which can be overcome by the use of alternative nickel-containing phases. We investigated here crystalline nickel carbide nanoparticles (NPs) as a catalyst in a colloidal suspension for hydrogenation reactions under H2 (7 bar) and below 100 °C. The Ni3C NP catalyst was prepared by thermal decomposition of Ni(acac)2 at 250 °C in a mixture of oleylamine and 1-octadecene, and characterized by X-ray diffraction (XRD) on powder, transmission electron microscopy (TEM) and X-ray absorption spectroscopy (XAS). Polar solvents appeared comparatively more favorable than less polar ones for the hydrogenation of two model substrates: nitrobenzene and phenylacetylene. Furthermore, the presence of water in the solvent mix was mostly favorable to the hydrogenation yield. Considering the known metastability of Ni3C at elevated temperatures (>300 °C), TEM, XRD, XAS and X-ray photoelectron spectroscopy (XPS) were used to verify the structural integrity of the Ni3C phase and the evolution of the nanoparticle surface upon catalysis. Complementarily, we confirmed that the nanoparticles are the active catalytic species and not leached species. Lastly, we expanded the scope to a variety of aldehydes, ketones, esters, nitriles and unsaturated hydrocarbons. Altogether, this study highlights the relevance of Ni3C, a catalytic phase so far overlooked for these hydrogenation reactions.


 Please wait while we load your content...
Please wait while we load your content...