Insight into the regulation between crystallinity and oxygen vacancies of BiVO4 affecting the photocatalytic oxygen evolution activity†
Abstract
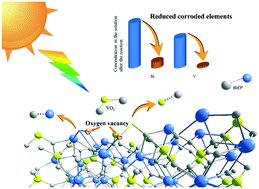
The surface defect structure and material crystallinity determine the catalytic activity and stability of the photocatalyst. In this work, decahedral BiVO4 nanoparticles with a uniform size are synthesized by the solution-chemistry method. The effects of different heat treatment temperatures on their morphology, size, crystallinity and photocatalytic oxygen evolution activity were systematically studied. The morphology and structure characterization show that with the increase of heat treatment temperature, the size and crystallinity of BiVO4 nanoparticles increase significantly, while the proportion of defective oxygen decreases. The photocatalytic oxygen evolution tests show that although the oxygen evolution activity of the as-prepared BiVO4 without heat treatment is the highest, the photocorrosion phenomenon is aggravated by the defective structure. Inductively coupled plasma (ICP) results show that photocorrosion is assigned to the reduction and dissolution of vanadium (VI) within BiVO4 by photogenerated electrons. Improving the material's crystallinity can effectively enhance the V–O bond energy and avoid the photocorrosion effect to a certain extent. The proposed BiVO4 calcined at 400 °C exhibits good stability and catalytic activity for catalytic oxygen evolution. Under visible light (λ > 420 nm) irradiation, the photocatalytic oxygen evolution rate in Fe(NO3)3 solution is 149.3 μmol h−1. In Fe2(SO4)3 solution, the oxygen evolution rate is 95.4 μmol h−1, corresponding to an AQY of 37.1% at 420 nm. The experimental results show that NO3− ions also play the role of electron scavengers.


 Please wait while we load your content...
Please wait while we load your content...