Highly enhanced photocatalytic hydrogen evolution activity by modifying the surface of TiO2 nanoparticles with a high proportion of single Cu atoms†
Abstract
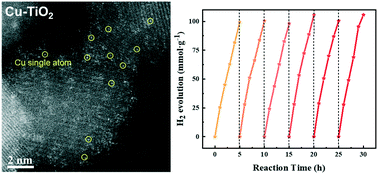
Single-atom photocatalysts have received a lot of attention owing to their high catalytic activity. It is vital that single atoms are highly dispersed on the surface of the catalyst for them to contribute their activity. However, the number of highly dispersed single atoms on the surface is limited by the agglomeration at higher loading concentrations or by the small specific surface area of the catalyst. In this study, TiO2 nanoparticles modified with single Cu atoms were synthesized by hydrothermal method with a high specific surface area (158.23 m2 g−1) and thus enhanced the exposure rate of single Cu atoms. The Cu–TiO2 exhibited a higher hydrogen evolution rate (21.053 mmol g−1 h−1) than other TiO2-based photocatalysts in previous reports and showed good stability after a long-term catalysis test (the hydrogen evolution efficiency barely decreased in a 30 hour catalytic reaction and in a 84-day long-term catalytic test). Moreover, the optimized sample exhibits an apparent quantum efficiency of 13.61% at 365 nm. The study builds the foundation to design the advanced material for excellent photocatalytic activity as well as durability.


 Please wait while we load your content...
Please wait while we load your content...