Synergetic effect between Pd2+ and Ir4+ species promoting direct ethane dehydrogenation into ethylene over bimetallic PdIr/AC catalysts†
Abstract
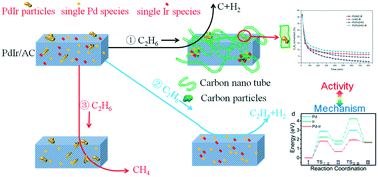
This work reported an efficient PdIr/activated carbon (PdIr/AC) catalyst and its application during direct ethane dehydrogenation (EDH) into ethane. The XRD and HADDF-STEM results revealed Pd/Ir nano-particles and atomically dispersed Pd2+ and Ir4+ species simultaneously. The activity tests demonstrated that Ir species showed higher C2H4 selectivity than Pd species and the co-presence of Pd/Ir could further improve the catalytic stability and inhibit carbon deposition over the bimetallic Pd/Ir catalyst. Nevertheless, raising the reaction temperature from 500 °C to 600 °C boosted coking including carbon nanotubes (CNTs) and carbon particles. XPS results of Pd7Ir2/AC-B after Ar or H2 pretreatment proved that the active sites are Pd2+/Ir4+ species. The simulation results exposed the reaction pathway over Ir4+/Pd2+ species, and ethane dehydrogenation was the rate-determining step over the PdIr/AC sample; meanwhile, the synergetic effect between Pd2+ and Ir4+ increased their TOF (C2H4). Finally, a deactivation mechanism was also proposed to examine the coking over various metal species.


 Please wait while we load your content...
Please wait while we load your content...