Promotional effect of Sn additive on the chlorine resistance over SnMnOx/LDO catalysts for synergistic removal of NOx and o-DCB†
Abstract
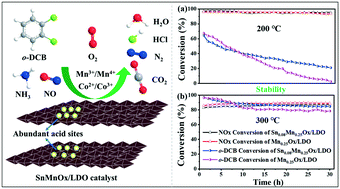
The chlorine poisoning of manganese-based catalysts is a critical issue to be solved for synergistic decomposition of nitrogen oxide (NOx) and chlorinated volatile organic compounds (Cl-VOCs). Herein, a series of SnMnOx/LDO catalysts were prepared by introducing Sn additive using a co-precipitation method, and exhibit higher activity and chlorine resistance than the MnOx/LDO catalysts. The characterization results indicate that the catalysts modified by Sn have smaller particle size, larger specific surface area, better structural stability and more abundant acid sites. The main reason for the improved chlorine resistance of the catalyst is that Sn introduces more B acid sites, which promotes the desorption of Cl species from the catalysts surface in the form of HCl. Moreover, the interactions between NH3-SCR and o-dichlorobenzene (o-DCB) oxidation over the Sn0.08Mn0.25Ox/LDO catalyst are different at high or low temperature. At low temperature, the oxidation of o-DCB is inhibited due to the competitive adsorption of o-DCB and NH3 on acid sites. At high temperature, NH3-SCR and o-DCB oxidation exhibit a synergistic effect. The o-DCB inhibits the excessive oxidation of NH3 and NO, and NO2 generated in situ from NO is conductive to the oxidation of o-DCB. This work is expected to provide the experimental and theoretical basis for the practical application of manganese-based catalysts in the field of simultaneous degradation of NOx and Cl-VOCs.


 Please wait while we load your content...
Please wait while we load your content...