Heparosan oligosaccharide synthesis using engineered single-function glycosyltransferases†
Abstract
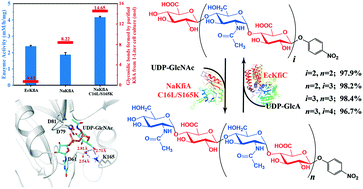
Synthetic towards homogeneous heparin oligosaccharides are increasing in terms of the number of products and the synthetic scale. The dual-function glycosyltransferase Pasteurella multocida heparosan synthase 2 (PmHS2) is widely used in these synthetic approaches. However, the unwanted degradation (caused by multi-step reverse-glycosylation) action of PmHS2 limits the catalytic efficiency of the process and the purity of the products, especially when the target oligosaccharide is longer than a hexasaccharide. Here, we found that the degradation is due to the coexistence of two catalytic domains in PmHS2, which suggested that an approach employing single-function glycosyltransferases could overcome this problem. By screening members of the KfiA family, we obtained a novel single-function heparosan: α1-4-N-acetylglucosaminyltransferase from Neisseria animaloris, which had an excellent recombinant expression level in Escherichia coli. Then, multiple rounds of structure-guided site-directed mutagenesis of the enzyme, named NaKfiA, were performed, producing an NaKfiA C16L/S165K double-mutant with drastically improved enzyme activity and stability compared with wild-type NaKfiA, but retaining the excellent production level and substrate promiscuity. NaKfiA(C16L/S165K) purified from the cell lysate of 1 liter of fermented E. coli produced 14.65 mmol α1,4-linkage. Combining NaKfiA(C16L/S165K) with the single-function glucuronyltransferase EcKfiC from E. coli K5, size-defined heparosan oligosaccharides were accessible with 96–98% yield in sequential enzymatic reactions, while unwanted oligosaccharide byproducts were minimized. Overall, our study resulted in an efficient engineered biocatalyst and established a biocatalytic approach for size-controlled synthesis of heparosan oligosaccharides relying on two single-function glycosyltransferases.
- This article is part of the themed collection: Biocatalysis: A cross-journal collection


 Please wait while we load your content...
Please wait while we load your content...