Comparison of the catalytic performance of Au/TiO2 prepared by in situ photodeposition and deposition precipitation methods for CO oxidation at room temperature under visible light irradiation†
Abstract
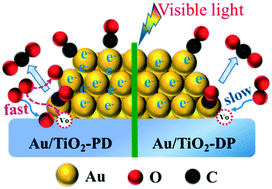
A Au/TiO2-PD sample was prepared via a mild in situ photodeposition method followed by heat treatment. As compared to Au/TiO2-DP prepared by deposition precipitation, this Au/TiO2-PD sample owned the smaller sizes of Au nanoparticles (Au NPs), and exhibited a better catalytic activity for CO oxidation under visible light irradiation or otherwise. The results of XPS, in situ EPR and in situ DRIFTS tests indicated that TiO2 could transfer electrons to Au NPs caused the increase in surface electron density of Au NPs, resulting in the enhanced adsorption of CO at Au sites. This adsorbed CO could interact with the lattice O of TiO2 to form CO2 accompanied by oxygen vacancy, while O2 could be adsorbed at the oxygen vacancy to form the lattice O of TiO2 again. Moreover, the other oxygen atom of O2 adsorbed at oxygen vacancy may form activated O− species at adjacent Au sites, which can further react with another CO adsorbed at Au sites to form CO2. As compared with Au/TiO2-DP, the Au/TiO2-PD sample showed more electron transfer from TiO2 to Au sites (meaning more CO activation) and then the more activation of O2, resulting in an ultra-high activity of CO oxidation. Further, under visible light, the Au/TiO2-PD sample also exhibited a more obvious photopromoted effect due to the higher surface electron density of Au NPs induced by LSPR effect than Au/TiO2-DP. This study showed that the effect of Au NPs size on CO oxidation may somewhat be attributed to the electron transfer behavior.


 Please wait while we load your content...
Please wait while we load your content...