Synthesis of gold nanocrystals with chiral morphology, chiral ligand and more exposed high-index facets as electrocatalyst for the oxidation of glucose enantiomers with high enantioselectivity and catalytic activity†
Abstract
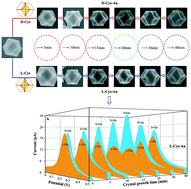
The poor enantioselectivity and catalytic activity of chiral metal nanocrystals limit their wide applications in chiral synthesis. In this study, we report a strategy for the synthesis of chiral gold nanocrystals via the reduction of HAuCl4 with ascorbic acid in the presence of CTAC, KBr and KI to form trioctahedral gold nanocrystals and their asymmetric structural evolution induced by D/L-cysteine. The resulting gold nanocrystals offer unique structures with a chiral morphology, chiral ligand and more exposed high-index facets. The formed D-Cys-Au and L-Cys-Au under optimized conditions exhibited high enantioselectivity and catalytic activity for the catalytic oxidation of glucose enantiomers. The catalytic activity of D-Cys-Au for L-glucose was 3.1-fold higher than that for D-glucose. The catalytic activity of L-Cys-Au for D-glucose was 3.4-fold higher than that for L-glucose. The DPV current of D-Cys-Au linearly increased and the DPV current of L-Cys-Au linearly decreased with an increase in the L-glucose/D-glucose ratio. The catalytic behavior of gold nanocrystals highly depend on the chiral morphology and chiral ligand. L-Cys-Au prepared with an evolution time of 20 min exhibited the best enantioselectivity. The increase in the coverage of L-cystine on L-Cys-Au resulted in reduced catalytic activity but improved enantioselectivity. The introduction of L-ascorbic acid hardly affected the catalytic activity of L-Cys-Au for D-glucose but obviously decreased the catalytic activity for L-glucose.


 Please wait while we load your content...
Please wait while we load your content...