Gas-phase equilibrium molecular structures and ab initio thermochemistry of anthracene and rubrene†
Abstract
Semi-experimental gas-phase structures of anthracene and rubrene (5,6,11,12-tetraphenyltetracene) were determined by means of gas electron diffraction (GED). The use of the flexible restraints in the refinement of the GED data successfully resolves non-equivalent C–C bond lengths. The tetracene core of an isolated rubrene molecule was found to exhibit a twist distortion of about 18°; this is less than DFT calculations predict (30–40°). The modified Feller–Peterson–Dixon method in conjunction with high-level DLPNO-CCSD(T) calculations was employed to resolve the discrepancy between the available experimental gas-phase enthalpies of formation for rubrene. The theoretical value of 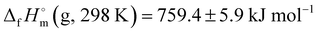 meets its recent experimental counterpart (765.6 ± 8.4 kJ mol−1) and is in strong disagreement with the previous estimation (882 kJ mol−1).
meets its recent experimental counterpart (765.6 ± 8.4 kJ mol−1) and is in strong disagreement with the previous estimation (882 kJ mol−1).



 Please wait while we load your content...
Please wait while we load your content...