Intrinsic fluorescence from firefly oxyluciferin monoanions isolated in vacuo†
Abstract
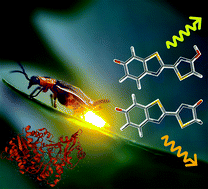
Fireflies, click beetles, and railroad worms glow in the dark. The color varies from green to red among the insects and is associated with an electronically excited oxyluciferin formed catalytically by the luciferase enzyme. The actual color tuning mechanism has been, and still is, up for much debate. One complication is that oxyluciferin can occur in different charge states and isomeric forms. We present here emission spectra of oxyluciferin monoanions in vacuo at both room temperature and at 100 K recorded with a newly developed and unique mass-spectroscopy setup specially designed for gas-phase ion fluorescence spectroscopy. Ions are limited to the phenolate-keto and phenolate-enol forms that account for natural bioluminescence. At 100 K, fluorescence band maxima are at 599 ± 2 nm and 563 ± 2 nm for the keto and enol forms, respectively, and at 300 K about 5 nm further to the red. The bare-ion spectra, free from solvent effects, serve as important references as they reveal whether a protein microenvironment redshifts or blueshifts the emission, and they serve as important benchmarks for nontrivial excited-state calculations.


 Please wait while we load your content...
Please wait while we load your content...