Hydrogen bond redistribution effects in mixtures of protic ionic liquids sharing the same cation: non-ideal mixing with large negative mixing enthalpies†
Abstract
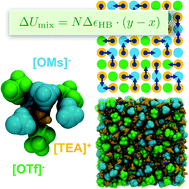
We report a joint experimental and theoretical study characterising the hydrogen bond (HB) redistribution in mixtures of two different protic ionic liquids (PILs) sharing the same cation: triethylammonium–methanesulfonate ([TEA][OMs]) and triethylammonium–trifluoromethanesulfonate ([TEA][OTf]). The mixing behaviour deviates strongly from ideality, exhibiting large negative energies of mixing. In the PIL, the [TEA] cation acts as a HB donor, being able to donate a single HB. Both, the [OMs] and the [OTf] anions can act as HB acceptors, which can accept multiple HBs via their respective SO3-groups. We use a combination of molecular dynamics (MD) simulations, calorimetry, and 1H-NMR chemical shift measurements to determine the difference in HB strength between the two species to be about 13 kJ mol−1, favouring the [TEA]–[OMs] interaction. Based on our MD simulations we are able to formulate a lattice model, discriminating between HB and nonspecific intermolecular interactions. We demonstrate that, due to the ordered structure of the PILs, mostly the HB interactions contribute to the mixing energy. This allows to us to connect the equilibrium of HBs to each of the two anion species with the mixing energies by a simple relation, which is obeyed by both, MD-simulation as well as experimental calorimetry and 1H-NMR chemical shift data.
- This article is part of the themed collection: 2022 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...