Denaturant- or ligand-induced changes in protein volume by pressure shift assay
Abstract
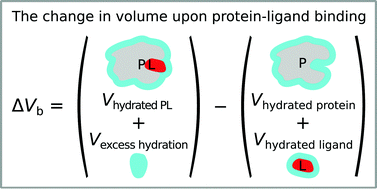
A complete thermodynamic description of protein–ligand binding includes parameters related to pressure and temperature. The changes in the protein volume and compressibility upon binding a ligand are pressure-related parameters that are often neglected due to the lack of routine methods for their determination. Fluorescent pressure shift assay (FPSA) is based on pressure-induced protein unfolding and its stabilization by a ligand and offers a universal approach to determine protein–ligand binding volumes. Extremely high pressures are required to unfold most proteins and protein–ligand complexes. Thus, guanidinium hydrochloride (GdmHCl) is used as a protein-destabilizing agent. We determined that GdmHCl unfolds carbonic anhydrase isoforms in a different pathway, but the destabilization effect is linear in a particular concentration range. We developed a concept for the FPSA experiment, where both – the ligand and GdmHCl – concentrations are varied. This approach enabled us to determine protein–ligand binding volumes that otherwise would be impossible due to the equipment-unreachable pressures of protein unfolding.


 Please wait while we load your content...
Please wait while we load your content...