Identification of incommensurability in l-leucine: can lattice instabilities be considered as general phenomena in hydrophobic amino acids?†
Abstract
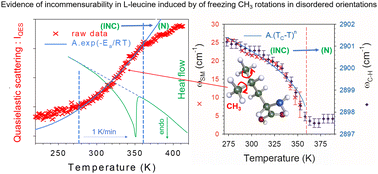
L-Leucine is an essential amino acid which has been focusing a lot of investigations on its phase transition sequence for more than fifty years. Combining Raman spectroscopy and X-ray diffraction experiments provides a new interpretation of the second order phase transition extending between 270 and 360 K as a displacive incommensurate-normal phase transition. A soft mode was clearly detected from low-frequency Raman investigations which exhibits the temperature dependence (A·(TC−T)1/2) typical of the temperature behavior of the amplitudon, an excitation specific to incommensurate phases. Simultaneously to the softening of the amplitudon, several very weakly intense X-ray reflections vanish upon heating at 360 K, and thereby are interpreted as satellite reflections. This incommensurability was described as resulting from the freezing of thermally activated hydrophobic side-chain rotations upon cooling in disordered orientations. Raman investigations were also performed on the isomeric amino acid L-norleucine previously identified as undergoing a normal-incommensurate phase transition around 200 K. Comparison of both studies suggests that the temperature behavior of thermally activated local motions generates lattice instabilities. Loss of periodicity can result from the freezing of rotations of molecular moieties in disordered orientations, or from the enhancement of anharmonicity of these rotations. This could be a general phenomenon in hydrophobic amino acids with direct consequences on their applications in the life science area.


 Please wait while we load your content...
Please wait while we load your content...