Phthalocyanine reactivity and interaction on the 6H-SiC(0001)-(3 × 3) surface investigated by core-level experiments and simulations†
Abstract
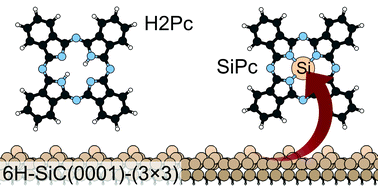
The adsorption of phthalocyanine (H2Pc) on the 6H-SiC(0001)-(3 × 3) surface is investigated using X-ray photoelectron spectroscopy (XPS), near edge X-ray absorption fine structure spectroscopy (NEXAFS), and density functional theory (DFT) calculations. Spectral features are tracked from the submonolayer to the multilayer growth regime, observing a significant modification of spectroscopic signals at low coverage with respect to the multilayer films, where molecules are weakly interacting. Molecules stay nearly flat on the surface at the mono and submonolayers. Previously proposed adsorption models, where the molecule binds by two N atoms to corresponding Si adatoms, do not reproduce the experimental spectra at the submonolayer coverage. We find instead that another adsorption model where the molecule replaces the two central H atoms by a Si adatom, effectively forming Si-phthalocyanine (SiPc), is both energetically more stable and yields in combination a better agreement between the experimental and simulated spectra. This suggests that the 6H-SiC(0001)-(3 × 3) surface may be a candidate substrate for the on-surface synthesis of SiPc molecules.
- This article is part of the themed collection: New Trends and Challenges in Surface Phenomena, Carbon Nanostructures and Helium Droplets


 Please wait while we load your content...
Please wait while we load your content...