Bipolar 1,8-naphthalimides showing high electron mobility and red AIE-active TADF for OLED applications†
Abstract
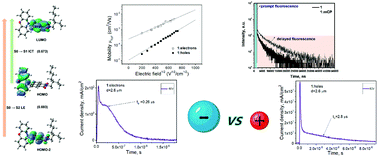
Aiming to design bipolar organic semiconductors with high electron mobility and efficient red thermally activated delayed fluorescence (TADF), three donor–acceptor compounds were designed and synthesized selecting 1,8-naphthalimide as an acceptor and phenoxazine, 3,7-di-tert-butylphenothiazine or 2,7-di-tert-butyldimethyl-9,10-dihydroacridine as donor moieties. Aggregation induced emission enhancement was detected for the compounds causing efficient TADF in the solid-state. Photoluminescence quantum yields up to 77% were observed for the films of the compounds doped in a host. The compounds exhibited small singlet–triplet splitting (0.03–0.05 eV), and high reverse intersystem crossing rates of 2.08 × 105–1.13 × 106 s−1. The compounds were characterized by satisfactory hole and electron-injecting properties with ionization potentials of 5.72–5.83 eV and electron affinities of 2.79–2.91 eV. Bipolar charge transport was revealed by time of flight measurements. Electron transport with low dispersity and mobilities exceeding 2 × 10−3 cm2 V−1 s−1 was observed at an electric field of 4.6 × 105 V cm−1. The compounds were used as emitters in red electroluminescent devices, which showed maximum external quantum efficiencies up to 8.2%. Utilization of host–guest systems as light-emitting materials with hosts preferably transporting holes and TADF guests which preferably transport electrons allowed maximum efficiencies to be achieved at a practical brightness of 700–2200 cd m−2. DFT calculations of the geometry, electronic structure, absorption and photoluminescence spectra of all compounds were carried out to prove the conclusions drawn from the experiment. The results of the calculations clearly show that the first excited state for all compounds is the intramolecular charge transfer state. Quantitative analysis of the separation degree of electronic density during excitation allows the observed dependence of the blue shift value in the absorption and emission spectra on the increasing polarity of the solvent to be explained.


 Please wait while we load your content...
Please wait while we load your content...