Recovery of syringic acid from aqueous solution by magnetic Fe–Zn/ZIF and its slow release from the CA-coated carrier based on the 3Rs concept†
Abstract
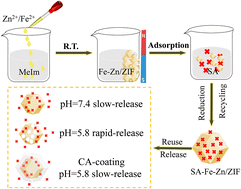
The excessive utilization of syringic acid (SA) has caused severe environmental pollution and economic waste. Herein, from an eco-friendly perspective, a magnetic nanomaterial (Fe–Zn/ZIF) was prepared by a one-step method at room temperature. Based on the 3Rs (pollutant reduction, resource recycling, and resource reuse) concept, this carrier exhibits great potential in the adsorption and release of SA. The adsorption process of SA over Fe–Zn/ZIF can reach equilibrium at ∼360 min with a high adsorption capacity of 450.6 mg g−1, superior to those of reported adsorbents, which can be ascribed to synergistic effects of strong electrostatic attraction, H-bonding interaction, and π–π interaction. To overcome the burst release of SA–Fe–Zn/ZIF in the weak acid phenomenon, calcium alginate (CA) was selected as an encapsulation material. As expected, CA@SA–Fe–Zn/ZIF only released 30.6% of SA in the first 180 minutes at pH = 5.8, a decrease of 14.8% compared to SA–Fe–Zn/ZIF. Therefore, the magnetic carrier obtained in this work may realize deep removal and controlled release of SA.
- This article is part of the themed collection: Editor’s Collection: Engineering Zeolitic Imidazolate Framework-8-based materials


 Please wait while we load your content...
Please wait while we load your content...