Non-covalent interactions of the hydroperoxo group in crystalline adducts of organic hydroperoxides and their potassium salts†
Abstract
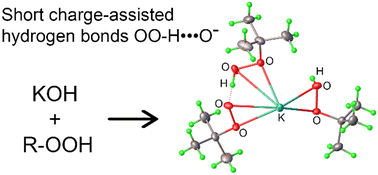
The present study demonstrates the similarity of hydrogen bonding of hydroperoxo functionality in organic hydroperoxide crystalline adducts (HPCA) and the much larger group of crystalline peroxosolvates. Based on a systematic analysis of the reported HPCA in the crystal database, the following common features, similar to those previously observed for crystalline peroxosolvates, could be derived: i) the hydroperoxo groups play a dominant structure-directing role in the crystal structure of HPCA; ii) organic hydroperoxides always form one hydrogen bond (per OOH group) as a proton donor (HB-D) in cocrystals; iii) a basic or amphoteric nature of the coformers is a requirement to obtain HPCA. Three new cocrystals of potassium salts of organic hydroperoxides with molecular hydroperoxides were synthesized. Single crystal X-ray diffraction revealed strong charge-assisted ROO−⋯HOOR hydrogen bonds (H-bonds). The energies/enthalpies of charge-assisted and ordinary H-bonds and K⋯O interactions in HPCA were estimated for the first time using solid-state DFT computations and periodic electron density analysis. The calculated energy values of the ordinary intermolecular HB-D are lower than those of the charge-assisted bonds and are close to those found for hydrogen peroxide in peroxosolvates. The H-bond energy values of hydroperoxo groups exceed those of the hydroxo groups in adducts with Cy3P![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O. Moreover, considering only the unique interactions, the total value of H-bond energies is almost twice as high as the total value of K⋯O coordination bond energies in one of the synthesized HPCA.
O. Moreover, considering only the unique interactions, the total value of H-bond energies is almost twice as high as the total value of K⋯O coordination bond energies in one of the synthesized HPCA.


 Please wait while we load your content...
Please wait while we load your content...