Isolation of unique heterocycles formed from pyridine-thiocarboxamides as diiodine, iodide, or polyiodide salts†
Abstract
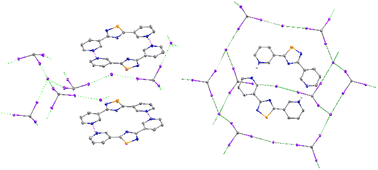
Given their two reactive centers, thioamides serve as useful synthetic building blocks for more complex heterocyclic molecules. The reaction of pyridine-4-thiocarboxamide (1) and pyridine-3-thiocarboxamide (2) with I2 provides protonated forms of the known products 3,5-di-(4-pyridyl)-1,2,4-thiadiazole (1A–1B) and 3,5-di-(3-pyridyl)-1,2,4-thiadiazole (2A–2C), respectively, and yielded new iodide and triiodide salts through the variation of reaction solvent. The analogous reaction of pyridine-2-thiocarboxamide (3) with I2 formed 2,4,6-tris(2-pyridinium)-1,3,5-triazine (3A) instead of a 1,2,4-thiadiazole containing product. The reaction of pyrazine-, pyrimidine-, and 5-methyl-pyridine-2-thiocarboxamide (4–6) in a variety of solvents yielded other heterocyclic products formed from the condensation of two, three, or four thioamide equivalents. Inclusion of bismuth triiodide in these reactions added an additional variable to manipulate, allowing for the further crystallographic study of these organic products. Four common BiI3-derived clusters, [Bi2I9]3−, [Bi4I16]4−, [Bi6I22]4−, and [Bi8I28]4−, were observed alongside these cationic heterocycles.


 Please wait while we load your content...
Please wait while we load your content...