Influence of water concentration on the solvothermal synthesis of VO2(B) nanocrystals†
Abstract
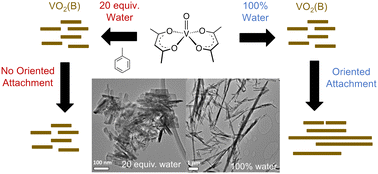
Nanocrystals of VO2(B) have attracted significant attention for their promising performance in electrochemical energy storage applications. Phase-purity and nanocrystal morphology are both crucial factors for the performance of these materials, but given the large number of crystal polymorphs available to VO2, achieving simultaneous control over crystal phase and nanocrystal size is a significant synthetic challenge. This paper describes the impact of water concentration on the synthesis of VO2(B) nanocrystals via solvothermal reaction of a molecular V(IV) precursor in toluene. Using controlled, stoichiometric amounts of water (20 equivalents per vanadium centre) enables access to short nanorods of VO2(B) whose lengths follow a Gaussian distribution with an average and standard deviation of 110 ± 30 nm. Decreasing the amount of water present to two or four equivalents results in formation of VO2(A) nanocrystals and eliminating it entirely results in no reaction. Increasing the amount of water to more than 20 equivalents increases the average length of the VO2(B) nanorods and causes the distribution in rod lengths to evolve from Gaussian to lognormal. This evolution in the size distribution is consistent with changes in a model Gaussian distribution observed upon simulated end-to-end oriented attachment events. These results demonstrate that control over the concentration of water is a useful strategy for tuning the morphology and crystal phase of VO2 nanocrystals.
- This article is part of the themed collection: Editor’s Collection: Non-classical crystallization processes


 Please wait while we load your content...
Please wait while we load your content...