Morphological reduction of Fe3O4 by a single-step hydrothermal synthesis using 1D MnO2 as a template and its supercapacitive behaviour
Abstract
Nanomaterials have attracted wide attention due to their promising characteristics in contrast to their bulk counterparts. However, techniques for their synthesis need to be explored further for their application in promising nanomaterial-based devices. Herein, a simple hydrothermal technique was developed for the synthesis of Fe3O4 nanoparticles utilizing nanoscale MnO2 as a template. It was found that the redox chemical conditions employed for the synthesis of nanostructured MnO2 are also applicable for the fabrication of Fe3O4 nanostructures. Subsequently, HRTEM, XRD and XPS analyses were conducted, which confirmed the presence of morphologically reduced Fe3O4. Furthermore, the electrochemical analysis revealed the promising supercapacitive behaviour of 590 F g−1 at a current density of 2.9 A g−1 for the as-synthesised MnO2/Fe3O4 nanocomposite. This high performance can be attributed to the favourable characteristics of the developed nanomaterial such as high specific surface area, abundant active sites and short diffusion paths for charges.
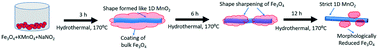


 Please wait while we load your content...
Please wait while we load your content...