Amorphous phase-mediated crystallization of calcium pyrophosphate tetrahydrate: the role of alkaline earth metal ions†
Abstract
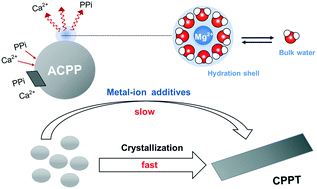
Although calcium pyrophosphates are commonly involved in crystal arthropathies, their formation mechanisms remain largely underexplored. Here, we investigated the crystallization pathway of calcium pyrophosphate tetrahydrate in the absence and presence of two biologically essential metal ions, Mg2+ and Sr2+, and another alkaline earth metal ion, Ba2+. The crystallization of calcium pyrophosphate tetrahydrate (CPPT) was found to proceed via a multistage process by first forming amorphous calcium pyrophosphate (ACPP), which then transformed into a crystalline phase. Alkaline earth metal ions can affect the process of CPPT crystallization by influencing the stability of the amorphous precursor and the subsequent crystal growth. Mg2+ exhibited the strongest effect on stabilizing ACPP, while Ba2+ showed a negligible effect on prolonging the lifetime of ACPP in solution. The surface adsorption of metal ions had a larger effect on the stability of ACPP than the bulk incorporation of metal ions. Interestingly, the ability of the metal-ion additives to inhibit ACPP crystallization was independent of their affinities to ACPP but correlated with their hydration abilities. These results suggest that the hydration characteristics of metal ions play more important roles in stabilizing ACPP than the content of adsorbed ions. These findings are relevant to understanding the pathogenesis of calcium pyrophosphate crystal deposition disease and the role of the metal ions involved in the process. This study also highlights the importance of the hydration characteristics of metal ions in regulating the stability of amorphous intermediate phases, providing a promising strategy for the precise control of biomineralization.


 Please wait while we load your content...
Please wait while we load your content...