Benzene and triazine-based porous organic polymers with azo, azoxy and azodioxy linkages: a computational study†
Abstract
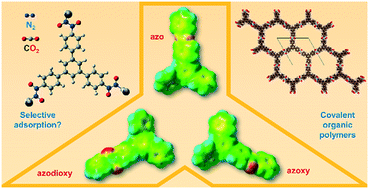
Among other porous materials, porous organic polymers have already been proven to be a valuable alternative for the selective adsorption of CO2 over N2. In a rational design of new porous organic systems, computational methods can help us to better understand structural features responsible for efficient and selective adsorption of gases. In this paper, a comprehensive approach relying on complementary computational chemistry methods including calculation of binding energies, visualization of electrostatic potential maps and using GCMC simulations to obtain adsorption isotherms and density plots is suggested for a detailed analysis of nitrogen–nitrogen linked porous organic systems. Three different linkages (azo, azoxy and azodioxy) and two different connectors (trisubstituted benzene and triazine) were computationally investigated referring to similar hexagonal porous organic structures. Considering the reversibility in azodioxy bond formation, azodioxy-based 2D layered structures, along with azoxy systems, seem to be new promising candidates for future development of porous organic materials capable of selective binding of CO2 over N2.


 Please wait while we load your content...
Please wait while we load your content...