Effects of alkylthio groups on phase transitions of organic molecules and liquid crystals: a comparative study with alkyl and alkoxy groups†
Abstract
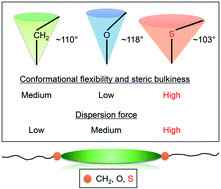
To describe the effects of the alkylthio groups on the phase transition behavior of organic liquid crystal (LC) molecules and their causes, we herein scrutinize the melting points (Tm) and nematic (N)–isotropic phase transition temperatures (TNI or TIN) of alkylthio-, alkoxy-, and alkyl-based LC homologues. To begin with, phase transitions are compared for three homologous series of 4-cyanophenyl benzoates bearing 4-alkyl-, 4-alkoxy-, and 4-alkylthio groups (denoted by nCPB, nOCPB, and nSCPB, respectively) with different alkyl chains (CnH2n+1; n = 1–8). In contrast to nCPB and nOCPB, some of which exhibit enantiotropic N phases, all alkylthio-based nSCPB are found to display monotropic N phases. The nSCPB homologues exhibit higher Tm than not only their corresponding nCPB for all n but also nOCPB counterparts in the case of long chains (n ≥ 6); a similar trend is confirmed also for the Tm values of the structurally similar biphenyl- and cyanobiphenyl-based homologues. Meanwhile, the TNI or TIN values of nSCPB and nCPB are lower than those of nOCPB for all n, and higher for nSCPB than for nCPB with the short chains (n = 1 and 2) but comparable with one another for the longer chains (n ≥ 3). In addition, our discussion addresses more anisotropic 1,4-phenylene bis(benzoate) LCs with alkylthio and alkoxy groups. Furthermore, single-crystal structures of three LC molecules with methylthio groups are examined. Several interatomic contacts around the sulfur atoms such as S⋯HC contacts are indicated, which differ for respective molecules. The present study draws the following conclusions. The alkylthio groups mostly play the role of decreasing the phase transition temperatures (especially TNI or TIN) and hindering LC formation for organic rod-like molecules, primarily due to conformational flexibility, steric bulkiness, and lower molecular anisotropy imposed by C–S–C. The sulfur atom contributes to intermolecular attractive interactions, which are reflected in phase transitions as spatially averaged phenomena (i.e., dispersion force and more polarizable π-systems). The influence of those interactions become more pronounced with lengthening alkyl chains.


 Please wait while we load your content...
Please wait while we load your content...