Phosphine-catalyzed regio- and stereo-selective hydroboration of ynamides to (Z)-β-borylenamides†
Abstract
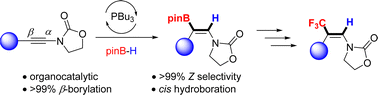
We report a tri-n-butyl phosphine catalyzed regio- and stereo-selective hydroboration of ynamides to yield (Z)-β-borylenamides in good yields. Surprisingly, a formal cis addition to the triple bond was observed as confirmed by NMR and X-ray crystallography. 31P NMR studies suggest that a zwitterionic vinylphosphonium intermediate is key in the mechanism. The resulting products were further transformed to β-CF3 enamides via stereoretentive trifluoromethylation.


 Please wait while we load your content...
Please wait while we load your content...