d–sp orbital hybridization: a strategy for activity improvement of transition metal catalysts
Abstract
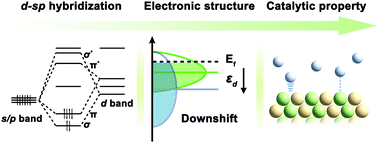
Orbital hybridization to regulate the electronic structures and surface chemisorption properties of transition metals has been extensively investigated for searching high-performance catalysts toward various reactions. Unlike conventional d–d hybridization, the d–sp hybridization interaction between transition metals and p-block elements could result in surprising electronic properties and catalytic activities. This feature article highlights the recent progress in the development of high-performance transition metal-based catalysts through the extraordinary d–sp hybridization strategy, particularly for energy-related electrocatalytic applications. We start by giving an introduction of fundamental concepts associated with electronic structures of transition metal catalysts, including the Sabatier principle, d-band theory, electronic descriptor, as well as the comparison of d–d hybridization and d–sp hybridization strategies. Then, we summarize the theoretical and experimental advances in d–sp hybridization catalysts, including p-block element-doped metal catalysts, intermetallic catalysts and supported metal catalysts, with emphasis on the important roles of d–sp hybridization in tuning catalytic performances. Finally, we present existing challenges and future development prospects for the rational design of advanced d–sp hybridization catalysts.
- This article is part of the themed collection: 2022 Pioneering Investigators


 Please wait while we load your content...
Please wait while we load your content...