Rhodium-catalyzed sequential B(3)-, B(4)-, and B(5)-trifunctionalization of o-carboranes with three different substituents†
Abstract
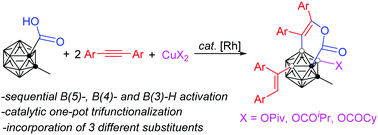
A rhodium-catalyzed one-pot trifunctionalization of o-carboranes with three different substituents via a carboxy group directed sequential B(5)-alkenylation, B(4)-alkyne annulation and B(3)-acyloxylation has been developed for the first time, leading to the synthesis of a new class of B(3,4,5)-trisubstituted o-carborane derivatives. Treatment of 1-COOH-2-CH3-o-C2B10H10 with ArC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CAr in the presence of a [Cp*RhCl2]2 catalyst and a Cu(OPiv)2 oxidant gave 1,4-[COOC(Ar)
CAr in the presence of a [Cp*RhCl2]2 catalyst and a Cu(OPiv)2 oxidant gave 1,4-[COOC(Ar)![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(Ar)]-2-Me-3-OPiv-5-[C(Ar)
C(Ar)]-2-Me-3-OPiv-5-[C(Ar)![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH(Ar)-o-C2B10H7 in good to high yields. This protocol represents a new strategy for the catalytic selective polyfunctionalization of carboranes with different substituents.
CH(Ar)-o-C2B10H7 in good to high yields. This protocol represents a new strategy for the catalytic selective polyfunctionalization of carboranes with different substituents.
- This article is part of the themed collection: Boron Chemistry in the 21st Century: From Synthetic Curiosities to Functional Molecules


 Please wait while we load your content...
Please wait while we load your content...