Inner residues of macrothiolactone in autoinducer peptides I/IV circumvent spontaneous S-to-O acyl transfer to the upstream serine residue†
Abstract
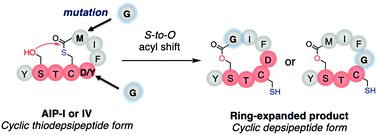
Autoinducing peptides I and IV (AIP-I/IV) are naturally occurring cyclic thiodepsipeptides (CTPs) bearing a Ser–Thr–Cys–Asp/Tyr (STC[D/Y]) tetrapeptide motif, where the Cys thiol (HSC) in the side-chain is linked to the Met C-terminal carboxylic acid (MCOOH) to form 5-residue macrothiolactones,−SC(D/Y)FIMCO−. We have recently reported that CTPs containing SX1CX2 motifs spontaneously undergo macrolactonization to yield cyclic depsipeptides (CDPs) by an unprecedented rapid S-to-O acyl transfer to the upstream Ser hydroxyl group. Interestingly, even though the STC[D/Y] motif in AIP-I/IV is a member of the SX1CX2 motif family, it maintains the CTP form. This suggests that AIP-I/IV have a structural or chemical motive for avoiding such an S-to-O acyl transfer, thus retaining the CTP form intact. Here we have used genetic code reprogramming to ribosomally synthesize various AIP-I analogs and studied what the determinant is to control the formation of CTP vs. CDP products. The study revealed that a Gly substitution of the inner Asp/Tyr or Met residues in the thiolactone drastically alters the resistance to the promotion of the S-to-O acyl transfer, giving the corresponding CDP product. This suggests that the steric hindrances originating from the α-substituted sidechain in these two amino acids in the AIP-I/IV thiolactone likely play a critical role in controlling the resistance against macrolactone rearrangement to the upstream Ser residue.
- This article is part of the themed collections: Synthesis and chemical biology of macrocycles and RSC Chemical Biology Transparent Peer Review Collection


 Please wait while we load your content...
Please wait while we load your content...