trans-Sialylation: a strategy used to incorporate sialic acid into oligosaccharides
Abstract
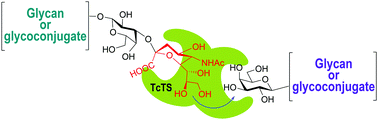
Sialic acid, as a component of cell surface glycoconjugates, plays a crucial role in recognition events. Efficient synthetic methods are necessary for the supply of sialosides in enough quantities for biochemical and immunological studies. Enzymatic glycosylations obviate the steps of protection and deprotection of the constituent monosaccharides required in a chemical synthesis. Sialyl transferases with CMP-Neu5Ac as an activated donor were used for the construction of α2-3 or α2-6 linkages to terminal galactose or N-acetylgalactosamine units. trans-Sialidases may transfer sialic acid from a sialyl glycoside to a suitable acceptor and specifically construct a Siaα2-3Galp linkage. The trans-sialidase of Trypanosoma cruzi (TcTS), which fulfills an important role in the pathogenicity of the parasite, is the most studied one. The recombinant enzyme was used for the sialylation of β-galactosyl oligosaccharides. One of the main advantages of trans-sialylation is that it circumvents the use of the high energy nucleotide. Easily available glycoproteins with a high content of sialic acid such as fetuin and bovine κ-casein-derived glycomacropeptide (GMP) have been used as donor substrates. Here we review the trans-sialidase from various microorganisms and describe their application for the synthesis of sialooligosaccharides.
- This article is part of the themed collection: Celebrating Latin American Chemistry


 Please wait while we load your content...
Please wait while we load your content...