A rapid and quantitative detection method for plasma soluble growth stimulating gene protein 2 based on time resolved fluorescence immunochromatography
Abstract
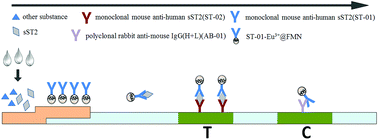
Background: plasma soluble growth stimulating gene protein 2 (sST2) is a new generation biomarker in heart failure (HF), which is an independent predictor of adverse outcomes of heart failure. Thus, the establishment of a rapid and sensitive method for detecting sST2 is urgently needed. Methods: lanthanide element Eu3+ coated fluorescent nanometer microspheres (Eu3+@FMN) can be used as markers to label monoclonal mouse anti-human sST2 antibody ST-01 (ST-01-Eu3+@FMN). When the immune sandwich complex formed between the monoclonal mouse anti-human sST2 antibody ST-02 and ST-01-Eu3+@FMN on the test band with the appearance of target object sST2, we can detect the fluorescence intensity of Eu3+ on the test band and the quality control band using a dry fluorescence analyzer. We calculated the T/C value (T/C = fluorescence intensity of the test band/fluorescence intensity of the quality control band), fitted to the calibration curve, and measured the concentration value of sST2 in the corresponding sample. Results: the best reaction time was 15 min after condition exploration, and the optimal sample volume was 80 μL. The detection sensitivity of the scheme was 2.14 ng mL−1. The calibration curve of the assay was y = 0.0113x + 0.0033, and the linear range was 5–200 ng mL−1. No cross reaction was found when the samples contained BNP, NT-proBNP, and galectin-3, indicating a good specificity. The precision was good with a relative deviation < 15%. The coefficient of variation of detection results of low-concentration samples and high-concentration samples was 4.20% and 3.30% respectively in the same batch of strip tests, so the intra-assay CV was set as <10%; when different batches of strips were used for testing, the coefficient of variation of detection results of low-concentration samples and high-concentration samples was 10.06% and 8.38% respectively, so the inter-assay CV was set as <15%. Stability test results showed that the relative deviation of test results at each time node was <15%, indicating good stability of the assay strips. The correlation coefficient between the ST-01-Eu3+@FMN based time-resolved fluorescence immunochromatography analysis and sST2 ELISA kit was 0.98. To confirm the usage of our proposed TRF-ICA for clinical samples, it was used to determine the concentration of sST2 in samples obtained from 34 patients with heart insufficiency, acute and chronic heart failure. As a result, we successfully detected a minimal concentration of 5.21 ng mL−1 and a maximum concentration of 184.26 ng mL−1 for sST2. Conclusion: this technique provides a rapid, simple and quantitative detection method for sST2 in clinics. It can help clinicians to predict the incidence of adverse events in patients with HF.


 Please wait while we load your content...
Please wait while we load your content...