Ratiometric fluorescence enzyme-linked immunosorbent assay based on carbon dots@SiO2@CdTe quantum dots with dual functionalities for alpha-fetoprotein
Abstract
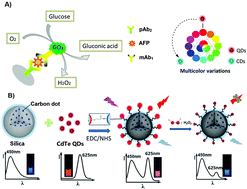
Molecular tags such as fluorophores are increasingly being replaced with nanoparticles thanks to their superior optical properties, substantial chemical stability, and stability against photobleaching. Herein, we innovatively constructed a new ratiometric fluorescence enzyme-linked immunosorbent assay (RF-ELISA) for the screening of alpha-fetoprotein (AFP) in early hepatocellular carcinoma in vitro diagnostics using carbon dots@SiO2@CdTe quantum dots (CDs@SiO2@CdTe QDs). Carbon dots with blue fluorescence were initially encapsulated into SiO2 nanospheres through the typical Stöber method. Thereafter, CdTe QDs with red fluorescence were modified onto the surface of CDs@SiO2 nanospheres. Dual-emission nanotags with blue and red fluorescent signals were utilized to design a RF-ELISA method for the determination of AFP on the anti-AFP capture antibody-coated microplate using glucose oxidase (GOx)-labeled anti-AFP secondary antibody. After the formation of the sandwiched immunocomplex, GOx catalyzed glucose to generate hydrogen peroxide (H2O2), which could quench the red fluorescence of CdTe QDs on the surface of nanotags. Meanwhile, the encapsulated carbon dots in the nanotags could still maintain the initial blue fluorescence intensity. The ratio between red fluorescence intensity and blue-emission intensity could be used for the quantitative monitoring of AFP concentration under optimum conditions. The experimental results indicated that CDs@SiO2@CdTe QDs-based RF-ELISA could exhibit a good fluorescence signal with a dynamic linear range of 0.05–60 ng mL−1 at a low detection limit of 8.7 pg mL−1. Moreover, the fluorescence color of the solution including CDs@SiO2@CdTe QDs changed from pink to purple to blue with the increasing AFP level when viewed by the naked eye. Good reproducibility, high specificity, and acceptable stability were achieved for the analysis of target AFP. Importantly, the accuracy of ratiometric fluorescence immunoassay was evaluated to determine human serum samples, giving well-matched results relative to commercially usable human AFP ELISA method.


 Please wait while we load your content...
Please wait while we load your content...