Online restricted access molecularly imprinted solid-phase extraction coupled with liquid chromatography-mass spectrometry for the selective determination of serum bile acids
Abstract
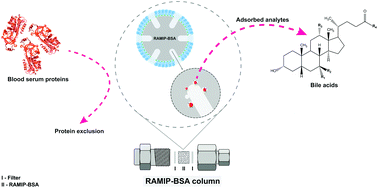
A rapid and sensitive online restricted access molecularly imprinted solid-phase extraction method coupled to a liquid chromatography-mass spectrometry (LC-MS) system for simultaneous determination of serum bile acids as well as their taurine and glycine conjugates was developed. Reversible liver damage of workers exposed to volatile organic solvents can be investigated based on the level of the analyzed molecules. A restricted access molecularly imprinted polymer coated with bovine serum albumin (RAMIP-BSA) was synthesized and used as the extraction phase. The column switching liquid chromatography system was able to exclude about 100% of the macromolecules and extract/separate nine bile acids from blood human serum samples, in a total time of 40 minutes. The developed method was validated based on the Food and Drug Administration (FDA) guidelines, being linear for all the analytes in their respective analytical ranges (coefficients of determination higher than 0.99), with limits of detection (LOD) and quantification (LOQ) ranging from 2.0 to 5.7 μg L−1 and from 10.0 to 25.0 μg L−1, respectively. Suitable results for precision (relative standard deviation ranged from 3.2 to 14.5% and 0.7 to 14.8%, respectively for intra and inter-assay) and accuracy (relative error ranged from −14.8 to 14.2%; −13.8 to 14.3%) were obtained. The validated analytical method was applied to determine bile acids in serum samples of five workers occupationally exposed to volatile organic solvent, demonstrating its applicability in the assessment of these toxicants.


 Please wait while we load your content...
Please wait while we load your content...