Thermally activated delayed fluorescence poly(dendrimer)s – detrapping excitons for reverse intersystem crossing†
Abstract
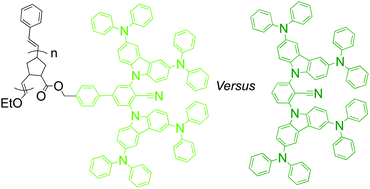
We report thermally activated delayed fluorescence (TADF) poly(dendrimer)s, composed of a norbornenyl-derived polymer backbone and dendritic side-chain chromophores comprising benzonitrile as the electron acceptor and two differently substituted first-generation 3,6-disubstituted carbazoles as electron donors. In particular, we compare the effect of the donor strength on the properties of the poly(dendrimer)s and their dendrimer analogues. The groups attached to the carbazolyl moieties to form the donor units were either 9,9-di-n-propylfluorene or diphenylamine. All four compounds were found to have TADF emission with neat film photoluminescence quantum yields (PLQYs) of up to 27%. The di-n-propylfluorenyl groups were found to afford a higher degree of protection from intrapolymer interchromophore interactions along the poly(dendrimer) backbone, and the solution and neat film PLQYs were similar. Photoluminescence studies performed at 77 K revealed that the triplet excitons can migrate along the poly(dendrimer) chain until they reach a chromophore that has the configuration required for charge transfer emission. Also, neat films of the poly(dendrimer)s were found to transport holes and electrons with reasonably balanced mobilities, whilst the dendrimers only had measurable hole mobilities. Organic light-emitting diodes with neat and blended emissive layers were found to have modest performance.
- This article is part of the themed collection: Materials for thermally activated delayed fluorescence and/or triplet fusion upconversion


 Please wait while we load your content...
Please wait while we load your content...