The “burst effect” of hydrogen desorption in MgH2 dehydrogenation†
Abstract
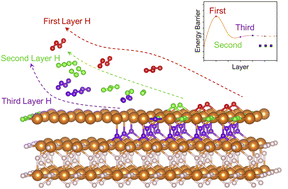
Magnesium hydride (MgH2) is a promising material for solid hydrogen storage due to its superior hydrogen storage capacity. However, its commercial application is inhibited by the sluggish dehydrogenation kinetics resulting from the complex hydrogen migration and desorption processes. Herein, we study the sequential MgH2 dehydrogenation mechanism by analyzing the kinetic and structural changes during the layer-by-layer hydrogen desorption process. Our results obtained by spin-polarized density functional theory calculations with van der Waals corrections (DFT-D3) unveiled an interesting “burst effect” during MgH2 dehydrogenation. We found that the initial dehydrogenation barriers (2.52 and 2.53 eV) are much higher than the subsequent reaction barriers (0.12–1.51 eV). The Mg–H bond analyses by the crystal orbital Hamilton population method indicate that the Mg–H bond strength decreases along the dehydrogenation process. Therefore, the subsequent H migration and hydrogen desorption become significantly easier, showing a “burst effect”. Electronic structure analyses using the electron localization function show that the H vacancy still has a high degree of electronic localization when the first layer of atomic H exists. Furthermore, ab initio molecular dynamics simulations were performed to analyze the kinetic characteristics of MgH2 after surface dehydrogenation to provide more evidence. This identified burst effect provides a theoretical basis for the dehydrogenation kinetics of MgH2 and proposes important guidelines for modifying MgH2-based hydrogen storage materials: promoting the initial dehydrogenation by structural engineering could be the key to facilitating the hydrogen desorption of MgH2.


 Please wait while we load your content...
Please wait while we load your content...